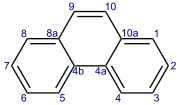
Phenanthrene
| |

| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
Phenanthrene | |
| Identifiers | |
3D model (JSmol)
|
|
| 1905428 | |
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.001.437 |
| EC Number |
|
| 28699 | |
| KEGG | |
| MeSH | C031181 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C14H10 | |
| Molar mass | 178.234 g·mol−1 |
| Appearance | Colorless solid |
| Density | 1.18 g/cm3[1] |
| Melting point | 101 °C (214 °F; 374 K)[1] |
| Boiling point | 332 °C (630 °F; 605 K)[1] |
| 1.6 mg/L[1] | |
| −127.9·10−6 cm3/mol | |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Flash point | 171 °C (340 °F; 444 K)[1] |
| Structure | |
| C2v[2] | |
| 0 D | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Phenanthrene is a polycyclic aromatic hydrocarbon (PAH) with formula C14H10, consisting of three fused benzene rings. It is a colorless, crystal-like solid, but can also appear yellow. Phenanthrene is used to make dyes, plastics, pesticides, explosives, and drugs. It has also been used to make bile acids, cholesterol and steroids.[3]
Phenanthrene occurs naturally and also is a man-made chemical. Commonly, humans are exposed to phenanthrene through inhalation of cigarette smoke, but there are many routes of exposure. Animal studies have shown that phenanthrene is a potential carcinogen.[3] However, according to IARC, it is not identified as a probable, possible or confirmed human carcinogen.[4]
Phenanthrene's three fused rings are angled as in the phenacenes, rather than straight as in the acenes. The compound with a phenanthrene skeleton and nitrogens at the 4 and 5 positions is known as phenanthroline.
History and etymology
[edit]Phenanthrene was discovered in coal tar in 1872 independently by Carl Graebe (article manuscript received on November 1st[5]) as well as by Wilhelm Rudolph Fittig and his doctoral student Eugen Ostermayer (manuscript received on November 19th[6] but Ostermayer's dissertation defended in August[7]). Fittig and Ostermayer were able to determine the structure of the compound by oxidizing it first to a corresponding quinone and then to diphenic acid, and soon Graebe confirmed it by a synthesis from stilbene.[8]
Prior to February 1873 Fittig sent a letter to Graebe where he proposed to name the hydrocarbon phenanthrene (German: Phenanthren) in order to account for its similarity to biphenyl and anthracene, which was swiftly adopted.[9]
Physical properties
[edit]Phenanthrene is nearly insoluble in water but is soluble in most low-polarity organic solvents such as toluene, carbon tetrachloride, ether, chloroform, acetic acid and benzene.
Phenanthrene is fluorescent under ultraviolet light, exhibiting a large Stoke shift.[10] It can be used in scintillators.
Chemistry
[edit]Reactions of phenanthrene typically occur at the 9 and 10 positions, including:
- Organic oxidation to phenanthrenequinone with chromic acid[11]
- Organic reduction to 9,10-dihydrophenanthrene with hydrogen gas and raney nickel[12]
- Electrophilic halogenation to 9-bromophenanthrene with bromine[13]
- Aromatic sulfonation to 2 and 3-phenanthrenesulfonic acids with sulfuric acid[14]
- Ozonolysis to diphenylaldehyde[15]
Canonical forms
[edit]Phenanthrene is more stable than its linear isomer anthracene. A classic and well established explanation is based on Clar's rule. A novel theory invokes so-called stabilizing hydrogen–hydrogen bonds between the C4 and C5 hydrogen atoms.[citation needed]
Synthesis
[edit]The Bardhan–Sengupta phenanthrene synthesis is a classic way to make phenanthrenes.[16]
This process involves electrophilic aromatic substitution using a tethered cyclohexanol group using diphosphorus pentoxide, which closes the central ring onto an existing aromatic ring. Dehydrogenation using selenium converts the other rings into aromatic ones as well. The aromatization of six-membered rings by selenium is not clearly understood, but it does produce H2Se.
Phenanthrene can also be obtained photochemically from certain diarylethenes (Mallory reaction):

Other synthesis routes include the Haworth reaction and the Wagner-Meerwein-type ring-expansion, as depicted below:

Commercially phenanthrene is not synthesized but extracted from the byproducts of coal coking, since it makes around 4–6% of coke oven coal tar.[17]
Natural occurrences
[edit]Ravatite is a natural mineral consisting of phenanthrene.[18] It is found in small amounts among a few coal burning sites. Ravatite represents a small group of organic minerals.
In plants
[edit]Phenanthrene derivatives occur in plants as phenanthrenoids. They have been reported from flowering plants, mainly in the family Orchidaceae, and a few in the families Dioscoreaceae, Combretaceae and Betulaceae, as well as in the lower plant class Marchantiophyta (liverworts).[19]
See also
[edit]References
[edit]- ^ a b c d e Record of CAS RN 85-01-8 in the GESTIS Substance Database of the Institute for Occupational Safety and Health
- ^ Peter Atkins, J. D. P., Atkins' Physical Chemistry. Oxford: 2010. P. 443.
- ^ a b "Phenanthrene Fact Sheet" (PDF). archive.epa.gov. U.S. Environmental Protection Agency. Retrieved 19 July 2019.
- ^ "Phenanthrene". Sigma-Alrdich.
- ^ Graebe, C. (1872). "Ueber einen neuen dem Anthracen isomeren Kohlenwasserstoff". Berichte der deutschen chemischen Gesellschaft. 5 (2): 861–863. doi:10.1002/cber.18720050279. ISSN 0365-9496.
- ^ Ostermayer, E.; Fittig, R. (1872). "Ueber einen neuen Kohlenwasserstoff aus dem Steinkohlentheer". Berichte der deutschen chemischen Gesellschaft. 5 (2): 933–937. doi:10.1002/cber.187200502100. ISSN 0365-9496.
- ^ Ostermayer, Eugen (1872). Ueber einen neuen Kohlenwasserstoff im Steinkohlentheeröl: Inaugural-Dissertation (in German). Druck v. Fues.
- ^ Graebe, C. (1873). "Ueber Synthese des Phenanthrens". Berichte der deutschen chemischen Gesellschaft. 6 (1): 125–127. doi:10.1002/cber.18730060147. ISSN 0365-9496.
- ^ Graebe, C. (1873). "Ueber das Verhalten der Chinone beim Erhitzen mit Natronkalk". Berichte der deutschen chemischen Gesellschaft. 6 (1): 63–66. doi:10.1002/cber.18730060124. ISSN 0365-9496.
- ^ "Spectrum [Phenanthrene] | AAT Bioquest". www.aatbio.com. Retrieved 2024-07-30.
- ^ Organic Syntheses, Coll. Vol. 4, p. 757 (1963); Vol. 34, p. 76 (1954).
- ^ Organic Syntheses, Coll. Vol. 4, p. 313 (1963); Vol. 34, p. 31 (1954).
- ^ Organic Syntheses, Coll. Vol. 3, p. 134 (1955); Vol. 28, p. 19 (1948).
- ^ Organic Syntheses, Coll. Vol. 2, p. 482 (1943); Vol. 16, p. 63 (1936).
- ^ Organic Syntheses, Coll. Vol. 5, p. 489 (1973); Vol. 41, p. 41 (1961).
- ^ "Bardhan Sengupta Synthesis". Comprehensive Organic Name Reactions and Reagents. Vol. 49. 2010. pp. 215–219. doi:10.1002/9780470638859.conrr049. ISBN 9780470638859.
- ^ Ma, Zhi-Hao; Wei, Xian-Yong; Liu, Guang-Hui; Liu, Fang-Jing; Zong, Zhi-Min (2021-05-15). "Value-added utilization of high-temperature coal tar: A review". Fuel. 292: 119954. doi:10.1016/j.fuel.2020.119954. ISSN 0016-2361.
- ^ Ravatite Mineral Data
- ^ Kovács, Adriána; Vasas, Andrea; Hohmann, Judit (2008). "Natural phenanthrenes and their biological activity". Phytochemistry. 69 (5): 1084–1110. Bibcode:2008PChem..69.1084K. doi:10.1016/j.phytochem.2007.12.005. PMID 18243254.
External links
[edit]- Phenanthrene at scorecard.org


